Description of Hydrocodone 10 325 mg
Hydrocodone 10 325 mg is one of the most common pain reliever medicines, prescribed by doctors for moderate to severe pain cases. It contains opioids (related to morphine). Thus the medicine can be habit-forming or addictive in nature. Hydrocodone is an over-the-counter medicine used to treat pain and inflammation. Moreover, Hydrocodone 10 325 mg has common use as an antitussive agent. This activity outlines the indications, mechanism of action, methods of administration, significant adverse effects, contraindications, monitoring, and toxicity of hydrocodone so that providers can direct patient therapy to optimal outcomes.
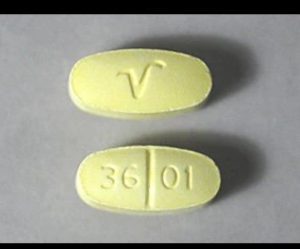
The shape of the Hydrocodone 10 325 mg pill
The shape of the Hydrocodone 10 325 mg pill is imprinted with V on top and 36 01 on the bottom of the tablet. The colour of the tablet is Yellow and the shape of the Tablet is Oblong and the generic name of the medicine is Hydrocodone/ Acetaminophen.
The picture of the pill of Valium 5 mg is given below.
Picture of the Valium 5 mg pill
Generic name: Hydrocodone/ Acetaminophen
Imprint: 36 01 V
Strength: 5 mg
Colour: Yellow
Size: 9.00 mm
Shape: Oblong
Availability: Prescription only
Dosage of Hydrocodone
Dosage of Hydrocodone for moderate to severe pain for Adult (ages 18 years and older and weighing at least 101 pounds [46 kg]) is 5 mg or 2.5 mg hydrocodone / 300 mg or 325 mg acetaminophen: The typical dosage is 1–2 tablets taken every 4–6 hours as needed. The maximum dosage is 8 tablets per day. 7.5 mg or 10 mg hydrocodone / 300 mg or 325 mg acetaminophen: The typical dosage is 1 tablet taken 4–6 hours as needed. The maximum dosage is 6 tablets per day.
Moreover, Buy Hydrocodone 10 325 mg online but is not recommended for children below 18 years old.
Side Effects of Hydrocodone
The severity of side effects varies from person to person and is influenced by a number of factors. As a result, you should only take this drug if your doctor has prescribed it after examining your medical situation thoroughly.
The majority of side effects fade away after 2-4 days or a week. Consult a doctor as soon as possible if it doesn’t go away. Some early symptoms arise and late symptoms arise as mentioned below:
Early symptoms
- Feeling drowsy
- Nausea and vomiting
- Slower than normal rates of breathing
- Low blood pressure
- Dry mouth
- Sleepiness
- Itchy skin
Later symptoms arise after a few days:
- Constipation (hard to have a bowel movement)
- Itching and hives
- Feeling drowsy or very sleepy
- Feeling dizzy
- Mood changes (feeling sadder or happier)
- Dry mouth
- Fainting
- Problems urinating
- Liver damage
- Blurry vision
- Confusion
Precautions And Warnings
- Repeated administration of opioids can lead to psychiatric dependency, physical dependence, and adaptation as a result, this product should be recommended and given with caution. When hydrocodone bitartrate and acetaminophen tablets are used for a short time to relieve pain, however, psychic dependence is unlikely to develop.
- Patients who take narcotics, antihistamines, antipsychotics, anti-anxiety medications, or other CNS depressants (including alcohol) with hydrocodone bitartrate and acetaminophen tablets may experience additive CNS depression.
- This medication may cause dizziness or drowsiness. You may become dizzy or sleepy if you consume alcohol. Do not drive, operate machinery, or engage in any other activity that requires attention until you are sure you can do so safely. Therefore avoid alcoholic beverages.
- This drug should only be taken during pregnancy unless absolutely necessary. It has the potential to harm an unborn child. Consult your doctor about the dangers and advantages. This medicine can transfer into breast milk and cause problems for a nursing baby.
- When hydrocodone and acetaminophen are taken together with CYP3A4 inhibitors like erythromycin, ketoconazole, and ritonavir, the hydrocodone plasma concentration may rise, resulting in greater or longer opioid effects.
- When hydrocodone and acetaminophen are taken together with CYP3A4 inducers such as rifampin, carbamazepine, and phenytoin, the plasma concentration of hydrocodone may drop, resulting in lower efficacy.
- Benzodiazepines or Other CNS Depressants: Combining hydrocodone and acetaminophen with benzodiazepines or other CNS depressants may raise the risk of hypotension, respiratory depression, severe sedation, coma, and death due to the additive physiological impact.
- Coadministration of hydrocodone and acetaminophen with other opioid analgesics (butorphanol, nalbuphine, pentazocine) may reduce the analgesic effect of opioids and/or trigger withdrawal symptoms.
- Taking hydrocodone and acetaminophen together with muscle relaxants may increase the neuromuscular blocking effect and cause more respiratory depression.
- The activation of antidiuretic hormone by the combination of hydrocodone and acetaminophen with other diuretics may diminish the efficacy of diuretics.
- Taking hydrocodone and acetaminophen at the same time as anticholinergic drugs might cause urinary retention and/or severe constipation.
Summary
Acetaminophen/hydrocodone is used in the treatment of back pain cough and belongs to the drug class narcotic analgesic combinations. Risk cannot be ruled out during pregnancy. Acetaminophen/hydrocodone 650 mg / 10 mg is classified as a Schedule 2 controlled substance under the Controlled Substance Act (CSA).
Moreover, Hydrocodone 10 325 mg is a painkiller in the opioid family (related to morphine). Acetaminophen is an over-the-counter medicine used to treat pain and inflammation. They may be combined in one prescription medicine to treat pain. An overdose occurs when someone takes more than the normal or recommended amount of this medicine.

Reviews
There are no reviews yet.